The No-Nonsense, Step-by-Step Guide to Getting Your CE Marking Declaration of Conformity
Look, you’ve poured your heart and soul into this product. It’s ready for the big leagues, right? But before you can slap that shiny CE marking on it and call it a day for the European market, there’s a Key hurdle. You need that Declaration of Conformity. And let me tell you, it’s not just a formality; it’s your product’s passport to millions of customers. So, how do you actually get your hands on this all-important document? Forget the jargon; we’re cutting straight to the chase.
Think of it like this: your product is the star performer, and the Declaration of Conformity is its official biography, proving it meets all the Needed requirements. It’s your sworn statement, your guarantee to consumers and authorities that your gizmo is safe, sound, and compliant. Sounds simple enough, but the devil, as always, is in the details. This isn’t a quick weekend project; it requires careful planning and execution.
Understanding What CE Marking Really Means

Source : scilife.io
First things first, let’s clear the air. CE marking isn’t some quality seal from a third-party agency. Nope. It’s a self-declaration by the manufacturer. You’re basically saying, “Yep, this thing meets all the EU’s health, safety, and environmental protection standards.” It’s your commitment. And frankly, it’s mandatory for a huge range of products sold within the European Economic Area (EEA). Ignoring it? That’s a fast track to getting your products yanked off shelves.
The Core Principle: Product Safety and Compliance
At its heart, the CE marking process is all about ensuring your product doesn’t pose a risk. Whether it’s a toy that shouldn’t have choking hazards, an electronic device that won’t electrocute someone, or machinery that won’t lop off a finger, the EU has directives for that. These directives lay out the Needed requirements – the absolute must-haves for safety and performance. Your Declaration of Conformity is the formal proof that you’ve ticked all those boxes.
Who is Responsible? Manufacturer vs. Importer
This is where it gets a bit tricky for some. Primarily, the manufacturer is the one on the hook for the CE marking and the Declaration of Conformity. If you build the product, you’re the one who needs to Make sure it complies. But what if you’re importing something from outside the EU? Then, the importer steps up. You become responsible for ensuring the product placed on the EU market is compliant. That means you need to verify that the manufacturer outside the EU actually did their homework.
Handling the EU Directives: It’s Not One-Size-Fits-All
Here’s a Key point: there isn’t just one CE marking directive. Oh no, that would be too easy. Different product categories fall under different EU directives. Think toys, machinery, medical devices, electronics – they all have their own specific rulebooks. You absolutely must identify the correct directive (or directives!) that apply to your specific product. Get this wrong, and your entire process is a waste of time and money.
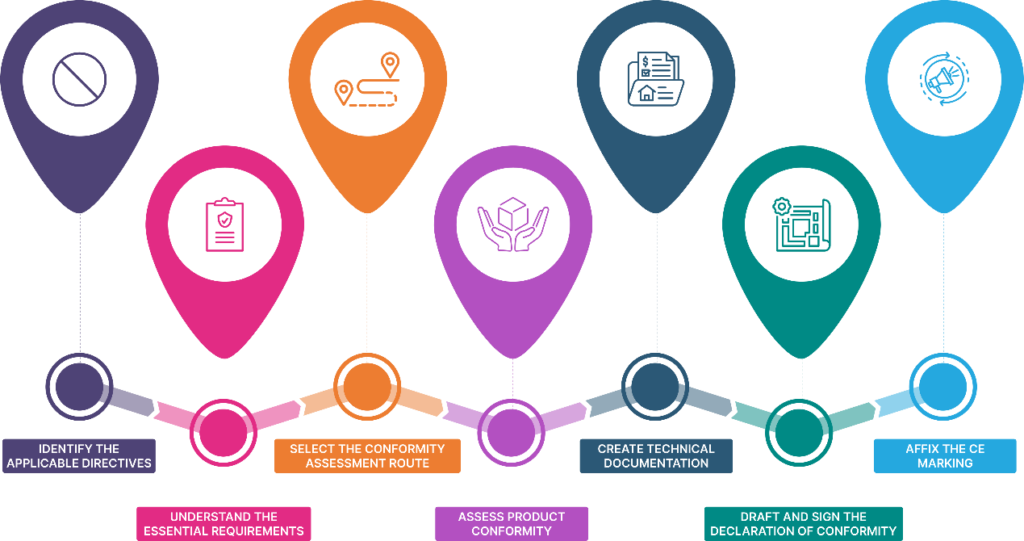
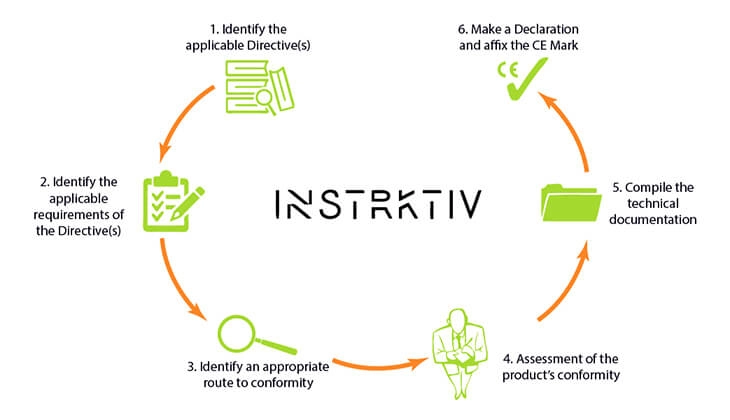
Step 1: Pinpointing the Applicable EU Directives
This is your foundational step. Seriously, don’t skip it. You need to be laser-focused on which EU directives govern your product. This isn’t guesswork. It involves researching your product type and understanding its intended use. A simple consumer gadget might fall under the Low Voltage Directive (LVD) and the Electromagnetic Compatibility (EMC) Directive. A power tool? That’s likely the Machinery Directive, plus potentially LVD and EMC. The wrong directive means the wrong requirements, and that’s a recipe for disaster.
Source : international.cliniexperts.com
Researching Your Product Category
Start by looking at your product. What is it? What does it do? Who is it for? Then, dive into the official EU websites or consult with experts. You’re looking for directives that specify Needed health and safety requirements. For example, if you’re making a children’s toy, the Toy Safety Directive is non-negotiable. If it’s a radio transmitter, the Radio Equipment Directive (RED) is key. This research phase is critical; get it wrong, and everything else crumbles.
Identifying Needed Requirements
Once you’ve identified the correct directive(s), you need to understand the Needed requirements they contain. These are the core safety and performance benchmarks your product must meet. They’re usually listed in the directives themselves, often in annexes. These requirements cover everything from materials used and electrical safety to noise emissions and mechanical hazards. You need to know them intimately.
Consulting the Notified Bodies List (If Applicable)
Some directives require the involvement of a Notified Body – an independent organization designated by an EU country to assess conformity. Not all products need this, but many higher-risk items do (like certain medical devices or construction products). Knowing if your product falls into a category requiring Notified Body involvement is part of this initial directive identification. You can find lists of Notified Bodies on the official EU NANDO database.
Step 2: Determining Conformity Assessment Procedures
Okay, you know the rules (the directives). Now, how do you prove you’ve followed them? This is where the conformity assessment procedure comes in. EU directives specify different modules or routes for demonstrating compliance. These range from simple self-assessment by the manufacturer to mandatory involvement of a Notified Body for testing and certification. Choosing the right module is Key.
Self-Assessment vs. Third-Party Involvement
For many directives, especially those with lower risk, you might be able to perform a self-assessment. This means you, the manufacturer, are responsible for testing and documenting that your product meets the Needed requirements. For higher-risk products, or those covered by specific directives, you’ll likely need to engage a Notified Body. They will perform or oversee specific conformity assessment activities.
Understanding Different Assessment Modules
The EU uses a system of modules (Modules A through H) to define conformity assessment. Module A, for instance, is manufacturer’s internal production control (self-assessment). Module G involves unit verification, and Module H requires a full quality system assessment by a Notified Body. Each directive typically specifies which modules are acceptable. You need to select the module(s) appropriate for your product and the relevant directive(s).
The Role of Harmonized Standards
Harmonized standards are key players here. These are technical specifications developed by European standardization organizations (CEN, CENELEC, ETSI). If your product complies with relevant harmonized standards, it is presumed to meet the Needed requirements of the directive(s) they cover. Using these standards simplifies the conformity assessment process significantly. They provide a clear, recognized path to compliance.
Step 3: Conducting Product Testing and Verification
This is where the rubber meets the road. You need concrete proof that your product meets the Needed requirements. This usually involves product testing. Depending on the directives and the chosen conformity assessment procedure, this testing might be done in-house by your own lab, by an independent third-party lab, or supervised by a Notified Body.
In-House Testing and Validation
If your product falls under a self-assessment route (like Module A), you’ll likely conduct internal testing. This means setting up a testing regime that specifically addresses the Needed requirements outlined in the relevant directives and standards. It’s Key that your internal testing is thorough, documented, and capable of withstanding scrutiny.
Utilizing Third-Party Test Laboratories
Sometimes, even if self-assessment is allowed, you might prefer or need to use an accredited third-party test laboratory. These labs have the specialized equipment and expertise to perform specific tests accurately. Their reports provide objective evidence of your product’s compliance, adding weight to your Declaration of Conformity. Make sure the lab is accredited for the specific tests required.
Specific Testing Requirements Based on Directives
The type of testing depends heavily on the directive. For electronics, it might be EMC (Electromagnetic Compatibility) testing to Make sure it doesn’t interfere with other devices and isn’t unduly affected by interference. For machinery, it could involve functional safety tests, noise level measurements, and mechanical strength assessments. For toys, it’s about chemical composition, flammability, and small parts. Every directive has its own checklist.
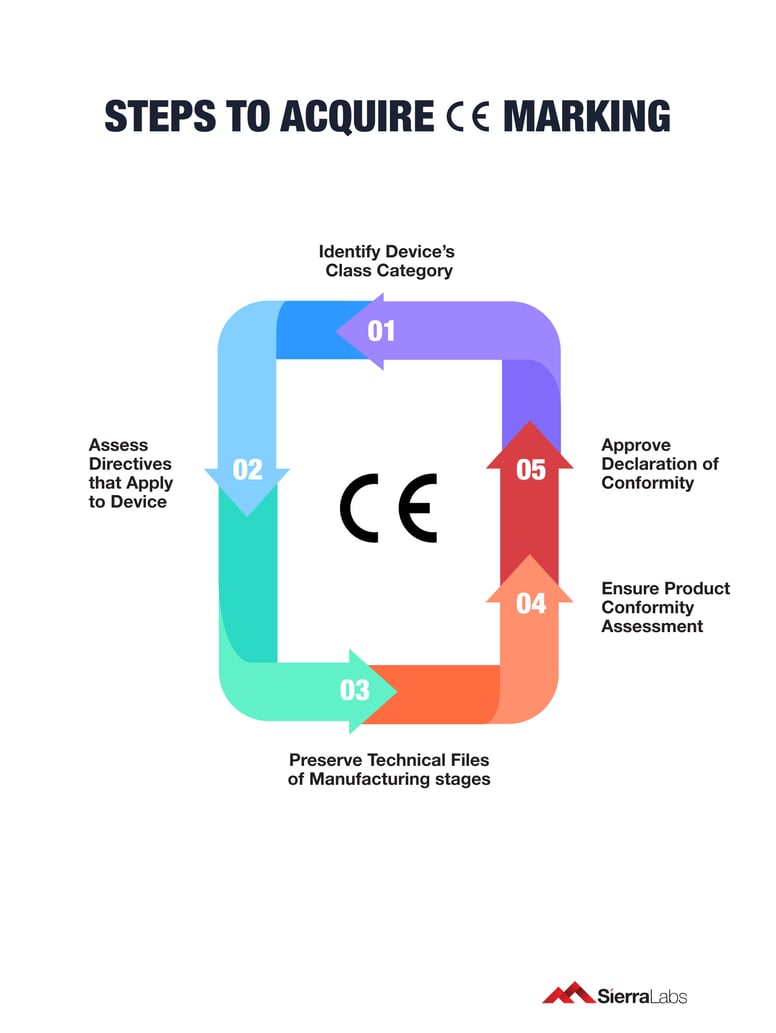
Step 4: Compiling the Technical Documentation

Source : iqmsglobal.com
This is arguably the most Large part of the process. The Technical Documentation (sometimes called a Technical File) is the Full record that proves your product complies with all applicable directives. It’s the evidence file you’d present if an authority ever asked to see it. It needs to be detailed, organized, and kept up-to-date for a specified period (often 10 years after the last product was placed on the market).
What Goes into the Technical File?
The technical documentation typically includes:
- A general description of the product.
- Engineering drawings, schematics, and component lists.
- The full text of the EU declarations of conformity of the components used.
- Reports from any tests performed (in-house or third-party).
- A list of the harmonized standards applied (or other technical specifications used).
- Details of the conformity assessment procedure followed.
- If a Notified Body was involved, their certificate.
- Risk assessment and the measures taken to Reduce identified risks.
Essentially, it tells the complete story of your product’s design, development, and verification against the regulations.
Risk Assessment: A Mandatory Component
Don’t underestimate the importance of the risk assessment. You need to systematically identify potential hazards associated with your product throughout its entire lifecycle – from manufacturing and transport to use and disposal. For each identified hazard, you must document the measures taken to eliminate or reduce the risk to an acceptable level. This demonstrates due diligence.
Keeping Documentation Up-to-Date
The technical documentation isn’t a one-and-done thing. If you make any changes to the product’s design, components, or manufacturing process that could affect its compliance with the Needed requirements, you must update the technical documentation accordingly. Keeping this file current is Key for ongoing compliance.
Step 5: Drafting the Declaration of Conformity (DoC)
Now for the star of the show – the Declaration of Conformity itself. This is the formal, written statement by the manufacturer (or their authorized representative) that the product meets all the applicable EU legal requirements. It’s a legal document, so accuracy and completeness are Top. Think of it as your product’s official birth certificate for the EU market.
Needed Information to Include
A typical Declaration of Conformity must contain specific information:
- Manufacturer’s name and full address (and authorized representative, if applicable).
- The product’s unique identification (e.g., serial number, model number, batch number).
- A statement that the declaration is issued solely under the responsibility of the manufacturer.
- Details of all applicable EU directives and regulations with which the product complies.
- Reference to any harmonized standards or other technical specifications used.
- The name and signature of the authorized person, along with their position.
- The date of issue.
- If a Notified Body was involved in the conformity assessment, their name, identification number, and details of the EU-type examination certificate.
Accuracy here is non-negotiable. Any errors can invalidate the declaration.
Using the Correct Wording
There’s often specific wording required by the directives for the DoC. You can’t just wing it. Make sure you are using the exact language prescribed by the relevant EU legislation. Templates are available, but always cross-reference them with the specific directive requirements to Make sure full compliance. This isn’t the place for creative writing.
Language Requirements for the DoC
The Declaration of Conformity must be drawn up in one of the official languages of the EU. Crucially, it must be in a language understood in the Member State where the product is being placed on the market or made available. Often, manufacturers provide it in the language of the destination country, alongside English or another widely understood language.
Step 6: Affixing the CE Marking
Once you have the signed Declaration of Conformity and all the supporting technical documentation is in order, you can finally affix the CE marking to your product. This visual symbol is your signal to the world that your product meets EU standards. It’s the culmination of all your hard work.
Placement and Visibility of the Mark
The CE marking must be clearly visible, legible, and indelible on the product itself. If this isn’t possible or practicable due to the nature of the product, it must be affixed to the packaging and to the accompanying documents (if any are required by the directive). Never apply the mark if you haven’t completed all the previous steps and issued the DoC.
Rules About the CE Marking’s Appearance
There are specific rules governing the appearance of the CE marking. It generally consists of the letters ‘CE’ in a specific proportion. If you need to enlarge or reduce the marking, you must maintain the proportions. If a Notified Body was involved in the production control phase, their identification number must also appear next to or below the CE marking.
Consequences of Misuse
Using the CE marking improperly is a serious offense. This includes applying it when it’s not required, using a similar-looking mark that could cause confusion, or applying it without having completed the conformity assessment and drawn up the Declaration of Conformity. Authorities can impose fines, order the product’s withdrawal from the market, or even ban its sale.
Source : blog.scalehealth.com
Step 7: Ongoing Compliance and Market Surveillance
Getting the Declaration of Conformity and affixing the CE mark isn’t the finish line. It’s more like the starting pistol. You need to Make sure that your product remains compliant throughout its production lifecycle and into the market. The authorities have the right to perform market surveillance to check if products actually meet the requirements.
Maintaining Technical Documentation
Before, your technical documentation must be kept for a specified period (often 10 years from the date the last product was manufactured). This is to allow authorities to check compliance retrospectively if needed. If you stop production, the clock doesn’t stop on your documentation obligations.
Handling Product Modifications
Any significant changes to the product design, materials, or manufacturing process that could impact its compliance with Needed requirements necessitate a review and potentially a re-assessment. You must Make sure that the Declaration of Conformity and the technical file still accurately reflect the product’s compliance status after modifications.
Responding to Market Surveillance Authorities
If market surveillance authorities investigate your product, you must be prepared to provide them with your Declaration of Conformity and the full technical documentation promptly. Failure to cooperate or provide the necessary evidence can lead to severe penalties, including market withdrawal and fines. Being organized and responsive is key.
Common Pitfalls and How to Avoid Them
Let’s be real, Handling this process can feel like walking through a minefield. Many businesses stumble on the same hurdles. Understanding these common mistakes is your best defense. Proactive planning and attention to detail are your best friends here.
Mistake 1: Applying the Wrong Directives
This is a big one. Misidentifying the applicable directives leads to fulfilling the wrong requirements, resulting in a non-compliant product and a faulty Declaration of Conformity. Always double-check which directives apply based on the product’s function, intended use, and potential risks.
Mistake 2: Incomplete or Incorrect Technical Documentation
Skipping steps, not performing thorough risk assessments, or failing to document testing properly are common errors. Remember, the technical file is your proof. It needs to be Full and accurate. If you can’t prove it, you haven’t done it. This is where many companies get tripped up during audits.
Mistake 3: Over-Reliance on Templates Without Understanding
Templates for Declarations of Conformity are useful, but they aren’t a magic bullet. You must understand the legal basis behind each piece of information required and Make sure it accurately applies to your specific product and the directives involved. A template filled out blindly is as good as useless.
Mistake 4: Assuming Compliance Without Proper Testing
Just believing your product is safe isn’t enough. You need objective evidence. Whether it’s in-house or third-party, rigorous testing is Needed. Don’t cut corners here; the safety of users and the legality of your product depend on it. Proper testing is non-negotiable.
Mistake 5: Not Keeping Documentation Current
Product design evolves. If these changes impact compliance, your documentation and Declaration of Conformity must be updated. Failing to do so can render your existing compliance invalid. Plan for updates and manage change control rigorously.
Example Scenario: The Smart Coffee Maker
Let’s imagine “BrewMaster Inc.” is launching a new smart coffee maker for the European market. It heats water, brews coffee, and connects via Wi-Fi to a smartphone app. This isn’t just a simple appliance; it’s got electrical components and connectivity.
Identifying Applicable Directives
BrewMaster needs to consider:
- Low Voltage Directive (LVD): Because it’s an electrical appliance operating within specific voltage ranges.
- Electromagnetic Compatibility (EMC) Directive: For its Wi-Fi connectivity and internal electronics to Make sure it doesn’t cause interference and isn’t susceptible to it.
- Radio Equipment Directive (RED): Specifically because it contains radio equipment (the Wi-Fi module).
- Potentially the RoHS Directive (Restriction of Hazardous Substances) if applicable to their components.
This requires careful checking of the specific scopes of each directive.
Source : instrktiv.com
Conformity Assessment and Testing
For LVD and EMC, they might be able to use self-assessment (Module A) if they have Strong internal testing capabilities. However, for the RED, due to the radio transmitter, they will likely need to involve a Notified Body to assess the radio aspects. They’ll need test reports for electrical safety, EMC, and the radio performance of the Wi-Fi module.
Technical Documentation and DoC
BrewMaster will compile a technical file including circuit diagrams, component lists, test reports from their lab and the Notified Body, risk assessments (e.g., risk of electric shock, fire hazard from heating element, secure Wi-Fi connection), and proof of compliance for all used components. Finally, they’ll draft the Declaration of Conformity, listing LVD, EMC, and RED, citing the harmonized standards used, and including the Notified Body’s details for the RED assessment. The Declaration will be provided in multiple languages for key markets.
Leveraging Professional Help: When to Call in the Experts
Look, not everyone has an in-house compliance department. And frankly, even if you do, sometimes the complexity requires external expertise. Bringing in specialists isn’t a sign of weakness; it’s a sign of smart business. They can save you time, money, and a whole lot of headaches.
Consultants Specializing in CE Marking
There are numerous CE marking consultants out there. These professionals live and breathe this stuff. They can help you identify the correct directives, guide you through the conformity assessment procedures, assist with technical documentation, and even help you find suitable Notified Bodies or testing labs. They are Crucial for complex products or if you’re new to the process.
Notified Bodies and Testing Laboratories
As discussed, these organizations are critical for certain product categories. Engaging with a Notified Body early in the process can clarify requirements and streamline the assessment. Similarly, reputable testing laboratories provide the objective data needed for your technical file and Declaration of Conformity. Choosing the right partners is Key.
Authorized Representatives
If you are an EU-based manufacturer, you handle your own CE marking. But if you are based outside the EU and want to appoint someone within the EU to act on your behalf, you can designate an Authorized Representative. This person or entity can handle communication with authorities and Make sure ongoing compliance, making market access smoother.
Getting your CE Marking Declaration of Conformity isn’t a walk in the park, but it’s absolutely achievable with the right approach. It requires diligence, a thorough understanding of the regulations, and Careful documentation. Don’t view it as a bureaucratic burden, but as a necessary step to Make sure your product is safe, legitimate, and ready to conquer the European market. Follow these steps, be prepared, and you’ll be well on your way.
Frequently Asked Questions
What exactly is a Declaration of Conformity (DoC)?
It's basically a legal document that says your product meets all the relevant EU safety, health, and environmental protection requirements. Like a CE marking promise.
Who is responsible for creating the DoC?
That would be the manufacturer or their authorized representative within the European Economic Area. You, or whoever made the product, have to take responsibility for its compliance.
What information needs to be included in the DoC?
Loads of stuff, really. Manufacturer's details, product description, list of directives/regulations applied, any harmonized standards used, and crucially, the signature of the person authorized. It needs to be thorough and accurate.
How often does the Declaration of Conformity need to be updated?
Whenever there are significant changes to the product, the directives it complies with, or the standards used. It's not a one-and-done thing; you gotta keep it current.
Do I need to submit the DoC to any authority?
Generally, no. You don't submit it to a central authority. However, you must be able to present it to market surveillance authorities if they ask for it. It’s for auditing purposes.